Sydney Parliament House, 09.07.2018
Cannabis has been greatly oversold by a left leaning press controlled by globalist and centralist forces while its real and known dangers have not been given appropriate weight in the popular press. In particular its genotoxic and teratogenic potential on an unborn generation for the next hundred years has not been aired or properly weighed in popular forums.
These weighty considerations clearly take cannabis out of the realm of personal choice or individual freedoms and place it squarely in the realm of the public good and a matter with which the whole community is rightly concerned and properly involved.
Cannabinoids are a group of 400 substances which occur only in the leaves of the Cannabis sativa plant where they are used by the plants as toxins and poisons in natural defence against other plants and against herbivores.
Major leading world experts such as Dr Nora Volkow, Director of the National Institute of Drug Abuse at NIH 1, Professor Wayne Hall, Previous Director of the Sydney Based National Drug and Alcohol Research Centre at UNSW 2, and Health Canada 3 – amongst many others – are agreed that cannabis is linked with the following impressive lists of toxicities:
1) Cannabis is addictive, particularly when used by teenagers
2) Cannabis affects brain development
3) Cannabis is a gateway to other harder drug use
4) Cannabis is linked with many mental health disorders including anxiety, depression,
psychosis, schizophrenia and bipolar disorder
5) Cannabis alters and greatly impairs the normal developmental trajectory – getting a
job, finishing a course and forming a long term stable relationship 4-11
6) Cannabis impairs driving ability 12
7) Cannabis damages the lungs
8) Cannabis is immunosuppressive
9) Cannabis is linked with heart attack, stroke and cardiovascular disease
10) Cannabis is commonly more potent in recent years, with forms up to 30% being widely available in many parts of USA, and oils up to 100% THC also widely available.
Serious questions have also been raised about its involvement in 12 different cancers, increased Emergency Room presentations and exposures of developing babies during pregnancy. It is with this latter group that the present address is mainly concerned.
Basic Physiology and Embryology Cells make energy in dedicated organelles called mitochondria. Mitochondrial energy, in the form of ATP, is known to be involved in both DNA protection and control of the immune system. This means that when the cell’s ATP is high DNA maintenance is good and the genome is intact. When cellular ATP drops DNA maintenance is impaired, DNA breaks remain unsealed, and cancers can form. Also immunity is triggered by low ATP.
As organisms age ATP falls by half each 20 years after the age of 20. Mitochondria signal and shuttle to the cell nucleus via several pathways. Not only do cells carry cannabinoid receptors on their surface, but they also exist, along with their signalling machinery, at high density on mitochondria themselves 13-19. Cannabis, and indeed all addictive drugs, are known to impair this cellular energy generation and thus promote the biochemical aging process 14-16,19,20. Most addictions are associated with increased cancers, increased infections and increased clinical signs of ageing 21-34.
The foetal heart forms very early inside the mother with a heartbeat present from day 21 of human gestation. The heart forms by complicated pathways, and arises from more than six groups of cells inside the embryo 35,36. First two arteries come together, they fold, then flex and twist to give the final shape of the adult heart. Structures in the centre of the heart mass called endocardial cushions grow out to form the heart valves between the atria and ventricles and parts of the septum which grows between the two atria and ventricles. These cardiac cushions, and their associated conoventricular ridges which grow into and divide the cardiac outflow tract into left and right halves, all carry high density cannabinoid type 1 receptors (CB1R’s) and cannabis is known to be able to interfere with their growth and development.CB1R’s appear on foetal arteries from week nine of human gestation 37.
The developing brain grows out in a complex way in the head section 35,36. Newborn brain cells are born centrally in the area adjacent to the central ventricles of the brain and then migrate along pathways into the remainder of the brain, and grow to populate the cortex, parietal lobes, olfactory lobes, limbic system, hypothalamus and hippocampus which is an important area deep in the centre of the temporal lobes where memories first form.
Developing bipolar neuroblasts migrate along pathways and then climb out along 200 million guide cells, called radial glia cells, to the cortex of the brain where they sprout dendrites and a major central axon which are then wired in to the electrical network in a “use it or lose it”, “cells that fire together wire together” manner.
The brain continues to grow and mature into the 20’s as new neurons are born and surplus dendrites are pruned by the immune system. Cannabinoids interfere with cellular migration, cellular division, the generation of newborn neurons and all the classes of glia, axonal pathfinding, dendrite sprouting, myelin formation around axons and axon tracts and the firing of both inhibitory and stimulatory synapses 14-16,19,20,38-40. Cannabinoids interfere with gene expression directly, via numerous epigenetic means, and via immune perturbation.
Cannabinoids also disrupt the mechanics of cell division by disrupting the mitotic spindle on which chromosomal separation occurs, causing severe genetic damage and frank chromosomal mis-segregation, disruption, rupture and pulverization 41-43.
Cannabis was found to be a human carcinogen by the California Environmental Protection agency in 2009 44. This makes it a likely human teratogen (deforms babies). Importantly, while discussion continues over some cancers, it bears repeating that a positive association between cannabis and testicular cancer was found in all four studies which investigated this question 45-49.
Cannabis Teratogenesis
The best animal models for human malformations are hamsters and rabbits. In rabbits cannabis exhibits a severe spectrum of foetal abnormalities when applied at high dose including shortened limbs, bowels hanging out, spina bifida and exencephaly (brain hanging out). There is also impaired foetal growth and increased foetal loss and resorption 50,51.
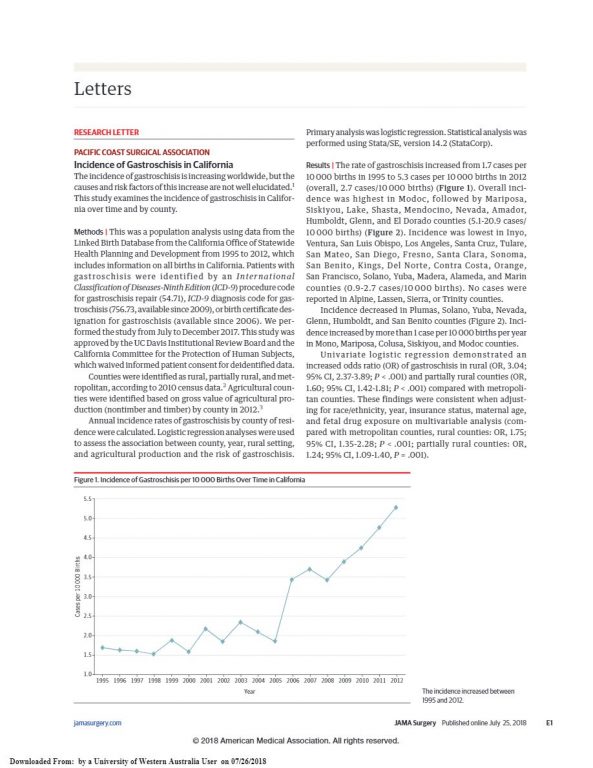
Many of these features have been noted in human studies 52. In 2014 Centres for Disease Control Atlanta Georgia reported increased rates of anencephaly (no brain, usually rapid death) gastroschisis (bowels hanging out), diaphragmatic hernia, and oesophageal narrowing 53,54. The American Heart Association and the American Academy of Pediatrics reported in 2007 an increased rate of ventricular septal defect and an abnormality of the tricuspid valve (Ebstein’s anomaly) 55. Strikingly, a number of studies have shown that cannabis exposure of the father is worse than that of the mother 56. In Colorado atrial septal defect is noted to have risen by over 260% from 2000-2013 (see Figure 1; note close correlation (correlation coefficient R = 0.95, P value = 0.000066) between teenage cannabis use and rising rate of major congenital anomalies in Colorado to 12.7%, or 1 in 8 live births, a rate four times higher than the USA national average !) 57.
And three longitudinal studies following children exposed to cannabis in utero have consistently noted abnormalities of brain growth with smaller brains and heads – persisting into adult life – and deficits of cortical and executive functioning persistent throughout primary, middle and high schools and into young adult life in the early 20’s 58-63. An Australian MRI neuroimaging study noted 88% disconnection of cortical wiring from the splenium to precuneus which are key integrating and computing centres in the cerebral cortex 38,39,64. Chromosomal defects were also found to be elevated in Colorado (rose 30%) 57, in Hawaii 52 in our recent analysis of cannabis use and congenital anomalies across USA, and in infants presenting from Northern New South Wales to Queensland hospitals 65. And gastroschisis shows a uniform pattern of elevation in all recent studies which have examined it (our univariate meta-analysis) 52,54,66-71.
Interestingly the gastroschisis rate doubled in North Carolina in just three years 1997-2001 72, but rose 24 times in Mexico 73 which for a long time formed a principal supply source for Southern USA 74. Within North Carolina gastroschisis and congenital heart defects closely followed cannabis distribution routes 74-76. In Canada a remarkable geographical analysis by the Canadian Government has shown repeatedly that the highest incidence of all anomalies – including chromosomal anomalies – occurs in those northern parts where most cannabis is smoked 77,78.
Congenital anomalies forms the largest cause of death of babies in the first year of life. The biggest group of them is cardiovascular defects. Since cannabis affects several major classes
of congenital defects it is obviously a major human teratogen. Its heavy epigenetic footprint,
by which it controls gene expression by controlling DNA methylation and histone modifications 79-81, imply that its effects will be felt for the next three to four generations – that is the next 100 years 82,83. Equally obviously it is presently being marketed globally as a major commodity apparently for commercial – or ideological – reasons. Since cannabis is clearly contraindicated in several groups of people including:
1) Babies
2) Children
3) Adolescents
4) Car drivers
5) Commercial Drivers – Taxis, Buses, Trains,
6) Pilots of Aeroplanes
7) Workers – Manual Tools, Construction, Concentration Jobs
8) Children
9) Adolescents
10) Males of Reproductive age
11) Females of Reproductive age
12) Pregnancy
13) Lactation
14) Workers
15) Older People – Mental Illness
16) Immunosuppressed
17) Asthmatics – 80% Population after severe chest infection
18) People with Personal History of Cancer
19) People with Family History of Cancer
20) People with Personal History of Mental Illness
21) People with Family History of Mental Illness
22) Anyone or any population concerned about ageing effects 34
… cannabis legalization is not likely to be in the best interests of public health.
Concluding Remarks
In 1854 Dr John Snow achieved lasting public health fame by taking the handle off the Broad Street pump and saving east London from its cholera epidemic, based upon the maps he drew of where the cholera cases were occurring – in the local vicinity of the Broad Street pump.
Looking across the broad spectrum of the above evidence one notices a trulyremarkable concordance of the evidence between:
1) Preclinical studies in
i) Rabbits and
ii) Hamsters
2) Cellular and biological mechanisms, particularly relating to:
i) Brain development
ii) Heart development
iii) Blood vessel development
iv) Genetic development
v) Abnormalities of chromosomal segregation
i. Downs syndrome
ii. Turners syndrome
iii. Trisomy 18
iv. Trisomy 13
vi) Cell division / mitotic poison / micronucleus formation
vii) Epigenetic change
viii) Growth inhibition
3) 84Cross-sectional Epidemiological studies, especially from:
i) Canada 77,85
ii) USA 86,87
iii) Northern New South Wales 65,88 4) Longitudinal studies from 58:
i) Ottawa 59-63
ii) Pittsburgh
iii) Netherlands
Our studies of congenital defects in USA have also shown a close concordance of congenital anomaly rates for 23 defects with the cannabis use rate indexed for the rising cannabis concentration in USA, and mostly in the three major classes of brain defects, cardiovascular defects and chromosomal defects, just as found by previous investigators in Hawaii 52.
Of no other toxin to our knowledge can it be said that it interferes with brain growth and development to the point where the brain is permanently shrunken in size or does not form at all. The demonstration by CDC twice that the incidence of anencephaly (no brain) is doubled by cannabis 53,54 implies that anencephaly is the most severe end of the neurobehavioural teratogenicity of cannabis and forms one end of a continuum with all the other impairments which are implied by the above commentary.
(Actually when blighted ova, foetal resorptions and spontaneous abortion are included in the teratological profile anencephaly is not the most severe end of the teratological spectrum – that is foetal death). It is our view that with the recent advent of high dose potent forms of cannabis reaching the foetus through both maternal and paternal lines major and clinically significant neurobehavioural teratological presentations will become commonplace, and might well become all but universal in infants experiencing significant gestational exposure.
One can only wonder if the community has been prepared for such a holocaust and tsunami amongst its children?
It is the view of myself and my collaborators that these matters are significant and salient and should be achieving greater airplay in the public discussion proceeding around the world at this time on this subject.
Whilst cannabis legalization may line the pockets of the few it will clearly not be in the public interest in any sense; and indeed the public will be picking up the bill for this unpremeditated move for generations to come. Oddly – financial gain seems to be one of the primary drivers of the present transnational push. When the above described public health message gets out amongst ambitious legal fraternities, financial gain and the threat of major medico-legal settlements for congenital defects – will quickly become be the worst reason for cannabis legalization.
Indeed it can be argued that the legalization lobby is well aware of all of the above concerns – and their controlled media pretend debate does not allow such issues to air in the public forum. The awareness of these concerns is then the likely direct reason that cannabis requires its own legislation. As noted in the patient information leaflet for the recently approved Epidiolex (cannabidiol oil for paediatric fits) the US Food and Drug Administration (FDA) is well aware of the genotoxicity of cannabinoids.
The only possible conclusion therefore is that the public is deliberately being duped. To which our only defence will be to publicize the truth.
Source: Summary of Address to Sydney Parliament House, 09.07.2018 by Professor Dr. Stuart Reece, Clinical Associate Professor, UWA Medical School. University of Western Australia
References
1 Volkow, N. D., Baler, R. D., Compton, W. M. & Weiss, S. R. B. Adverse Health Effects of Marijuana Use. New England Journal of Medicine 370, 2219-2227, doi:doi:10.1056/NEJMra1402309 (2014).
2 Hall, W. & Degenhardt, L. Adverse health effects of non-medical cannabis use. Lancet 374, 1383-1391, doi:10.1016/S0140-6736(09)61037-0 (2009).
3 Heath Canada. Health Effects of Cannabis, (2018).
4 Meier, M. H. et al. Associations Between Cannabis Use and Physical Health Problems in Early Midlife: A Longitudinal Comparison of Persistent Cannabis vs Tobacco Users. JAMA Psychiatry 73, 731-740,
doi:10.1001/jamapsychiatry.2016.0637 (2016).
5 Fergusson, D. M., Boden, J. M. & Horwood, L. J. Cannabis use and other illicit drug use: testing the cannabis gateway hypothesis. Addiction 101, 556-569, doi:ADD1322 [pii] 10.1111/j.1360-0443.2005.01322.x (2006).
6 Fergusson, D. M. & Horwood, L. J. Early onset cannabis use and psychosocial adjustment in young adults. Addiction 92, 279-296 (1997).
7 Fergusson, D. M., Horwood, L. J. & Beautrais, A. L. Cannabis and educational achievement. Addiction 98, 1681-1692, doi:573 [pii] (2003).
8 Fergusson, D. M., Horwood, L. J. & Swain-Campbell, N. Cannabis use and psychosocial adjustment in adolescence and young adulthood. Addiction 97, 1123- 1135, doi:103 [pii] (2002).
9 Fergusson, D. M., Lynskey, M. T. & Horwood, L. J. Conduct problems and attention deficit behaviour in middle childhood and cannabis use by age 15. The Australian and New Zealand journal of psychiatry 27, 673-682 (1993).
10 Lynskey, M. T., Fergusson, D. M. & Horwood, L. J. The origins of the correlations between tobacco, alcohol, and cannabis use during adolescence. Journal of child psychology and psychiatry, and allied disciplines 39, 995-1005 (1998).
11 Marie, D., Fergusson, D. M. & Boden, J. M. Links between ethnic identification, cannabis use and dependence, and life outcomes in a New Zealand birth cohort. The Australian and New Zealand journal of psychiatry 42, 780-788, doi:901464301 [pii] 10.1080/00048670802277289 (2008).
12 Fergusson, D. M., Horwood, L. J. & Boden, J. M. Is driving under the influence of cannabis becoming a greater risk to driver safety than drink driving? Findings from a longitudinal study. Accident; analysis and prevention 40, 1345-1350, doi:S0001- 4575(08)00026-2 [pii] 10.1016/j.aap.2008.02.005 (2008).
13 Bartova, A. & Birmingham, M. K. Effect of delta9-tetrahydrocannabinol on mitochondrial NADH-oxidase activity. J Biol Chem 251, 5002-5006 (1976).
14 Benard, G. et al. Mitochondrial CB(1) receptors regulate neuronal energy metabolism. Nat Neurosci 15, 558-564, doi:10.1038/nn.3053 (2012).
15 Hebert-Chatelain, E. et al. A cannabinoid link between mitochondria and memory. Nature 539, 555-559, doi:10.1038/nature20127 (2016).
16 Hebert-Chatelain, E. et al. Cannabinoid control of brain bioenergetics: Exploring the subcellular localization of the CB1 receptor. Mol Metab 3, 495-504, doi:10.1016/j.molmet.2014.03.007 (2014).
17 Koch, M. et al. Hypothalamic POMC neurons promote cannabinoid-induced feeding. Nature 519, 45-50, doi:10.1038/nature14260 (2015).
18 Mahoney, J. M. & Harris, R. A. Effect of 9 -tetrahydrocannabinol on mitochondrial precesses. Biochemical pharmacology 21, 1217-1226 (1972).
19 Wolff, V. et al. Tetrahydrocannabinol induces brain mitochondrial respiratory chain dysfunction and increases oxidative stress: a potential mechanism involved in cannabis-related stroke. Biomed Res Int 2015, 323706, doi:10.1155/2015/323706 (2015).
20 Chari-Briton, A. Proceedings: Swelling of rat liver mitochondria induced by delta1-tetrahydrocannabinol. Isr J Med Sci 11, 1189 (1975).
21 Reece, A. S. Chronic toxicology of cannabis. Clin Toxicol (Phila) 47, 517-524, doi:10.1080/15563650903074507 (2009).
22 Reece, A. S. Clinical implications of addiction related immunosuppression. J Infect 56, 437-445, doi:S0163-4453(08)00123-0 [pii] 10.1016/j.jinf.2008.03.003 (2008).
23 Reece A. S., Norman, A. & Hulse G.K. Acceleration of Cardiovascular – Biological Age by Amphetamine Exposure is a Power Function of Chronological Age. British Medical Journal Open (2017).
24 Reece A.S., Norman, A. & Hulse G.K. Cannabis Exposure as an Interactive Cardiovascular Risk Factor and Accelerant of Organismal Ageing – A Longitudinal Study. BMJ – Open 6(11) :e077891; doi:http://dx.doi.org/10.1136/bmjopen-2016- 011891 (2016).
25 Reece A.S. & G.K., H. Impact of Lifetime Opioid Exposure on Arterial Stiffness and Vascular Age: Cross-sectional and Longitudinal Studies in Men and Women. BMJ Open 4, 1-19, doi:10.1136/bmjopen-2013-004521 (2014).
26 Reece A.S. & Hulse G.K. Impact of Opioid Pharmacotherapy on Arterial stiffness and Vascular Ageing: Cross-sectional and Longitudinal Studies. Cardiovascular Toxicology 13, 254-266, doi:10.1007/s12012-013-9204-4 (2013).
27 Reece A. S. Relative and Age Dependent Stimulation of Soluble and Cellular Immunity in Opiate Dependence. Journal of Addiction Medicine 6, 10-17, doi:10.1097/ADM.0b013e31822c3bf4 (2012).
28 Reece A. S. Chronic Immune Stimulation as a Contributing Cause of Chronic Disease in Opiate Addiction Including Multi-System Ageing Medical hypotheses 75, 613-619, doi:10.1016/j.mehy.2010.07.047. Epub 2010 Aug 25. (2010).
29 Reece A.S. Improved Parameters of Metabolic Glycaemic and Immune Function and Arterial Stiffness with Naltrexone Implant Therapy. British Medical Journal, Case Reports (2008 ).
30 Reece A.S. & Hulse G. K.. Hypothalamic Pathophysiology in the Neuroimmune, Dysmetabolic and Longevity Complications of Chronic Opiate Dependency. J. Forensic Toxicology and Pharmacology 3, 3-46, doi:10.4172/2325-9841.1000126 (2014).
31 Reece, A. S. Evidence of Accelerated Ageing in Clinical Drug Addiction from Immune, Hepatic and Metabolic Biomarkers. Immun Ageing 4, 6-15, doi:10.1186/1742-4933-4-6. (2007).
32 Reece, A. S. Improved parameters of metabolic glycaemic and immune function and arterial stiffness with naltrexone implant therapy. BMJ Case Rep 2009, bcr0820080799, doi:10.1136/bcr.08.2008.0799 bcr08.2008.0799 [pii] (2009).
33 Reece, A. S. & Hulse, G. K. Elevation of the ACTH/cortisol ratio in female opioid dependent patients: A biomarker of aging and correlate of metabolic and immune activation. Neuro Endocrinol Lett 37, 325-336 (2016).
34 Reece A.S., Norman, A. & Hulse G.K. Cannabis Exposure as an Interactive Cardiovascular Risk Factor and Accelerant of Organismal Ageing – A Longitudinal Study. BMJ – Open 6, e011891-e011900, doi:http://dx.doi.org/10.1136/bmjopen- 2016-011891 (2016).
35 Sadler, T. W. Medical Embryology. 13th Edition edn, Vol. 1 1-406 (Wolters Kluwer, 2015).
36 Carlson, B. M. Human Embryology and Developmental Biology. Vol. 1 1-506 (Elsevier, 2014).
37 Pacher, P., Steffens, S., Hasko, G., Schindler, T. H. & Kunos, G. Cardiovascular effects of marijuana and synthetic cannabinoids: the good, the bad, and the ugly. Nat Rev Cardiol 15, 151-166, doi:10.1038/nrcardio.2017.130 (2018).
38 Lorenzetti, V. et al. Gross morphological brain changes with chronic, heavy cannabis use. Br J Psychiatry 206, 77-78, doi:10.1192/bjp.bp.114.151407 (2015).
39 Solowij, N. et al. Alteration to hippocampal shape in cannabis users with and without schizophrenia. Schizophrenia research 143, 179-184, doi:S0920-9964(12)00614-7 [pii] 10.1016/j.schres.2012.10.040 (2013).
40 Yates, D. Learning and memory: The cannabinoid connection. Nat Rev Neurosci 18, 4, doi:10.1038/nrn.2016.171 (2016).
41 Reece, A. S. & Hulse, G. K. Chromothripsis and epigenomics complete causality criteria for cannabis- and addiction-connected carcinogenicity, congenital toxicity and heritable genotoxicity. Mutat Res 789, 15-25, doi:10.1016/j.mrfmmm.2016.05.002 (2016).
42 Van Went, G. F. Mutagenicity testing of 3 hallucinogens: LSD, psilocybin and delta 9-THC, using the micronucleus test. Experientia 34, 324-325 (1978).
43 Zhang, C. Z. et al. Chromothripsis from DNA damage in micronuclei. Nature 522, 179-184, doi:10.1038/nature14493 (2015).
44 Tomar R.S. et al. Evidence on the Carcinogenicity of Marijuana Smoke. California Health Department 1, 1-150 (August 2009).
45 Callaghan, R. C., Allebeck, P., Akre, O., McGlynn, K. A. & Sidorchuk, A. Cannabis Use and Incidence of Testicular Cancer: A 42-Year Follow-up of Swedish Men between 1970 and 2011. Cancer Epidemiol Biomarkers Prev 26, 1644-1652,
doi:10.1158/1055-9965.EPI-17-0428 (2017).
46 Daling, J. R. et al. Association of marijuana use and the incidence of testicular germ cell tumors. Cancer 115, 1215-1223, doi:10.1002/cncr.24159 (2009).
47 Gurney, J., Shaw, C., Stanley, J., Signal, V. & Sarfati, D. Cannabis exposure and risk of testicular cancer: a systematic review and meta-analysis. BMC Cancer 15, 897, doi:10.1186/s12885-015-1905-6 (2015).
48 Lacson, J. C. et al. Population-based case-control study of recreational drug use and testis cancer risk confirms an association between marijuana use and nonseminoma risk. Cancer 118, 5374-5383, doi:10.1002/cncr.27554 (2012).
49 Trabert, B., Sigurdson, A. J., Sweeney, A. M., Strom, S. S. & McGlynn, K. A. Marijuana use and testicular germ cell tumors. Cancer 117, 848-853, doi:10.1002/cncr.25499 (2011).
50 Graham, J. D. P. in Cannabis and Health Vol. 1 (ed J.D.P. Graham) Ch. 8, 271-320 (Academic Press, 1976).
51 Geber, W. F. & Schramm, L. C. Effect of marihuana extract on fetal hamsters and rabbits. Toxicology and applied pharmacology 14, 276-282 (1969).
52 Forrester, M. B. & Merz, R. D. Risk of selected birth defects with prenatal illicit drug use, Hawaii, 1986-2002. Journal of toxicology and environmental health 70, 7-18 (2007).
53 van Gelder, M. M. et al. Maternal periconceptional illicit drug use and the risk of congenital malformations. Epidemiology 20, 60-66, doi:10.1097/EDE.0b013e31818e5930 (2009).
54 Van Gelder, M. M. H. J., Donders, A. R. T., Devine, O., Roeleveld, N. & Reefhuis, J. Using bayesian models to assess the effects of under-reporting of cannabis use on the association with birth defects, national birth defects prevention study, 1997-2005.
Paediatric and perinatal epidemiology 28, 424-433, doi:10.1111/ppe.12140 (2014).
55 Jenkins, K. J. et al. Noninherited risk factors and congenital cardiovascular defects: current knowledge: a scientific statement from the American Heart Association Council on Cardiovascular Disease in the Young: endorsed by the American Academy of Pediatrics. Circulation 115, 2995-3014,
doi:10.1161/CIRCULATIONAHA.106.183216 (2007).
56 Wilson, P. D., Loffredo, C. A., Correa-Villasenor, A. & Ferencz, C. Attributable fraction for cardiac malformations. Am J Epidemiol 148, 414-423 (1998).
57 Colorado: Department of Public Health and the Environment. Vol. 1 Birth Defect Data – Colorado Register of Congenital Surveillance Network 1 (ed Colorado: Department of Public Health and the Environment) http://www.chd.dphe.state.co.us/cohid/ (Colorado: Department of Public Health and the Environment, Denver Colorado, USA, 2018).
58 Brents L. in Handbook of Cannabis and Related Pathologies: Biology, Pharmacology, Diagnosis and Treatment Vol. 1 (ed Preedy V.R.) Ch. 17, 160-170 (Academic Press, 2017).
59 Fried, P. A. & Smith, A. M. A literature review of the consequences of prenatal marihuana exposure. An emerging theme of a deficiency in aspects of executive function. Neurotoxicol Teratol 23, 1-11 (2001).
60 Smith, A., Fried, P., Hogan, M. & Cameron, I. The effects of prenatal and current marijuana exposure on response inhibition: a functional magnetic resonance imaging study. Brain Cogn 54, 147-149 (2004).
61 Smith, A. M., Fried, P. A., Hogan, M. J. & Cameron, I. Effects of prenatal marijuana on response inhibition: an fMRI study of young adults. Neurotoxicol Teratol 26, 533-542, doi:10.1016/j.ntt.2004.04.004 (2004).
62 Smith, A. M., Longo, C. A., Fried, P. A., Hogan, M. J. & Cameron, I. Effects of marijuana on visuospatial working memory: an fMRI study in young adults. Psychopharmacology (Berl) 210, 429-438, doi:10.1007/s00213-010-1841-8 (2010).
63 Smith, A. M. et al. Prenatal marijuana exposure impacts executive functioning into young adulthood: An fMRI study. Neurotoxicol Teratol 58, 53-59, doi:10.1016/j.ntt.2016.05.010 (2016).
64 Zalesky, A. et al. Effect of long-term cannabis use on axonal fibre connectivity. Brain 135, 2245-2255, doi:aws136 [pii] 10.1093/brain/aws136 (2012).
65 Report of the Queensland Perinatal Maternal and Perinatal Quality Council & Queensland Health. Vol. 1 (ed Queensland Health) 5 (Queensland Health, Brisbane, 2018).
66 Werler, M. M., Sheehan, J. E. & Mitchell, A. A. Association of vasoconstrictive exposures with risks of gastroschisis and small intestinal atresia. Epidemiology 14, 349-354 (2003).
67 David, A. L. et al. A case-control study of maternal periconceptual and pregnancy recreational drug use and fetal malformation using hair analysis. PLoS One 9, e111038, doi:10.1371/journal.pone.0111038 (2014).
68 van Gelder, M. M. et al. Maternal periconceptional illicit drug use and the risk of congenital malformations. Epidemiology 20, 60-66, doi:10.1097/EDE.0b013e31818e5930 (2009).
69 Skarsgard, E. D. et al. Maternal risk factors for gastroschisis in Canada. Birth Defects Res A Clin Mol Teratol 103, 111-118, doi:10.1002/bdra.23349 (2015).
70 Torfs, C. P., Velie, E. M., Oechsli, F. W., Bateson, T. F. & Curry, C. J. A population-based study of gastroschisis: demographic, pregnancy, and lifestyle risk factors. Teratology 50, 44-53, doi:10.1002/tera.1420500107 (1994).
71 Draper, E. S. et al. Recreational drug use: a major risk factor for gastroschisis? Am J Epidemiol 167, 485-491, doi:10.1093/aje/kwm335 (2008).
72 Laughon, M. et al. Rising birth prevalence of gastroschisis. J Perinatol 23, 291-293, doi:10.1038/sj.jp.7210896 (2003).
73 Di Tanna, G. L., Rosano, A. & Mastroiacovo, P. Prevalence of gastroschisis at birth: retrospective study. BMJ (Clinical research ed 325, 1389-1390 (2002).
74 Centre, N. D. i. North Carolina Drug Threat Assessment, April 2003: Marijuana, (2003).
75 Nelson, J. S., Stebbins, R. C., Strassle, P. D. & Meyer, R. E. Geographic distribution of live births with tetralogy of Fallot in North Carolina 2003 to 2012. Birth Defects Res A Clin Mol Teratol 106, 881-887, doi:10.1002/bdra.23566 (2016).
76 Root, E. D., Meyer, R. E. & Emch, M. E. Evidence of localized clustering of gastroschisis births in North Carolina, 1999-2004. Social science & medicine (1982) 68, 1361-1367, doi:10.1016/j.socscimed.2009.01.034 (2009).
77 Leos-Toro C. et al. in Cannabis in Canada: Patterns and Trends, 2017 Vol. 1 (eds PROPEL, Centre for Population Health Impact, & University of Waterloo) 1-23 (University of Waterloo, Waterloo, Ontario, 2017).
78 Moore A., Roulean J. & Skarsgard E. Vol. 1 (ed Health Canada Public Health Agency of Canada) 57-63 (Health Canada, Ottawa, 2013).
79 DiNieri, J. A. et al. Maternal cannabis use alters ventral striatal dopamine D2 gene regulation in the offspring. Biol Psychiatry 70, 763-769, doi:10.1016/j.biopsych.2011.06.027 (2011).
80 Szutorisz, H. et al. Parental THC exposure leads to compulsive heroin-seeking and altered striatal synaptic plasticity in the subsequent generation. Neuropsychopharmacology 39, 1315-1323, doi:10.1038/npp.2013.352 (2014).
81 Watson, C. T. et al. Genome-Wide DNA Methylation Profiling Reveals Epigenetic Changes in the Rat Nucleus Accumbens Associated With Cross-Generational Effects of Adolescent THC Exposure. Neuropsychopharmacology, doi:10.1038/npp.2015.155 (2015).
82 Manikkam, M., Guerrero-Bosagna, C., Tracey, R., Haque, M. M. & Skinner, M. K. Transgenerational actions of environmental compounds on reproductive disease and identification of epigenetic biomarkers of ancestral exposures. PLoS One 7, e31901, doi:10.1371/journal.pone.0031901 (2012).
83 Manikkam, M., Tracey, R., Guerrero-Bosagna, C. & Skinner, M. K. Dioxin (TCDD) induces epigenetic transgenerational inheritance of adult onset disease and sperm epimutations. PLoS One 7, e46249, doi:10.1371/journal.pone.0046249 (2012).
84 Yang, X. et al. Histone modifications are associated with Delta9-tetrahydrocannabinol-mediated alterations in antigen-specific T cell responses. J Biol Chem 289, 18707-18718, doi:10.1074/jbc.M113.545210 (2014).
85 Public Health Agency of Canada. Vol. 1 (ed Health Canada Public Health Agency of Canada) 1-115 (Health Canada, Ottawa, 2013).
86 Substance Abuse and Mental Health Services Administration (SAMHSA), Department of Health and Human Services (HHS) & United States of America. National Survey on Drug Use and Health, (2018).
87 National Birth Defects Prevention Network. National Birth Defects Prevention Network, (2018).
88 Queensland Maternal and Perinatal Quality Council 2017. Vol. 1 1 (ed Queensland Health) 1-70 (Queensland Health, Brisbane, 2018).